
Introduction
Selecting the wrong o-ring material for food contact applications creates immediate regulatory exposure — product recalls, FDA warning letters, and contamination incidents that erode customer trust. FDA 21 CFR 177.2600 and NSF 61 compliance set the legal baseline for whether o-ring materials are acceptable in food, beverage, and potable water contact.
Many manufacturers assume that choosing "food-grade" materials guarantees compliance. In practice, proper specification requires understanding the distinction between FDA food contact regulations and NSF potable water standards, validating supplier certifications, and matching material properties to actual operating conditions — food type, temperature extremes, and aggressive CIP/SIP cleaning protocols.
Non-compliant materials introduce extractables that migrate into products, causing off-tastes, chemical contamination, and regulatory penalties that far exceed the cost of proper material selection.
This guide covers the technical and regulatory requirements for food-grade o-rings: compliant material families (EPDM, Silicone, FKM, NBR), application-specific selection criteria, testing protocols, and the documentation requirements that protect product safety and regulatory standing.
TL;DR
- FDA 21 CFR 177.2600 limits extractables in food-contact rubber; NSF 61 certifies materials for potable water safety
- EPDM excels in steam and aqueous foods; FKM handles fats and harsh chemicals; Silicone offers the widest temperature range; NBR provides economical oil resistance
- Compliance depends on validated certifications, migration testing, and lot traceability, not material selection alone
- Match material to food type, CIP/SIP peak temperatures, and cleaning agent compatibility
What FDA 21 CFR and NSF 61 Compliance Represents in Food Grade O-Rings
FDA 21 CFR 177.2600 establishes the regulatory framework for rubber compounds intended for repeated food contact. The regulation doesn't "approve" specific products — it lists permitted chemical ingredients (polymers, curing agents, antioxidants, fillers) and sets extraction limits based on food type.
For aqueous foods, total extractives must not exceed 20 mg/in² during the first 7 hours of testing, dropping to 1 mg/in² in subsequent testing. For fatty foods, limits are 175 mg/in² and 4 mg/in² respectively.
NSF 61 takes a different approach, focusing specifically on components in contact with potable drinking water. Rather than measuring mass migration, NSF 61 evaluates leachates against toxicological criteria to ensure no harmful substances exceed safe drinking water limits. Testing involves exposing materials to water under controlled conditions, then analyzing the water for contaminants using techniques like GC-MS and ICP-MS.
These three terms are not interchangeable — and confusing them creates real compliance risk:
- "Food contact safe" refers to FDA extraction limits for direct food contact materials
- "Food grade" broadly indicates suitability for food environments but carries no single standardized definition
- "Potable water safe" (NSF 61) addresses health-effects testing specific to drinking water systems

A material approved for dry food contact may fail in high-fat applications or hot water systems.
Compliance covers two layers: formulation (all ingredients must appear on the permitted substances list) and performance (migration testing under simulated use conditions). Both matter — and both are application-specific. Temperature, food type, and contact duration all determine whether a material holds up under actual operating conditions.
Factors That Influence Material Selection for Compliance
Regulatory compliance establishes the starting point, but real-world operating conditions determine which compliant material will actually perform reliably over time.
Food Type and Contact Conditions
Food chemistry dramatically affects material compatibility:
- Aqueous foods (water-based beverages, dairy, juices) are less aggressive extractors, making EPDM and Silicone viable choices
- Fatty foods (oils, butter, sauces, animal fats) are aggressive extractors requiring materials with low fat-soluble extractables—typically FKM or specially formulated EPDM grades
- Acidic foods (citrus juices, pickles, vinegar-based products) require materials resistant to pH extremes
- Alcoholic beverages demand materials that resist polar solvents without leaching flavor-altering compounds
Fatty foods present the greatest challenge because oils and fats extract substances more aggressively than water. Migration rates in fatty food simulants can run 10× higher than in aqueous simulants, which sharply limits material choices for applications like butter processing or cooking oil handling.
Temperature Exposure and Thermal Cycling
Temperature isn't just an operating parameter—it's a compliance variable. Migration rates increase exponentially with temperature. Studies on EPDM show that antioxidant migration at 100°C can be 2.78 times higher than at 40°C.
Critical temperature considerations include:
- Continuous operating temperature during production runs
- Peak temperature exposure during Steam-In-Place (SIP) sterilization cycles (often 250°F-280°F)
- Thermal shock from rapid heating during SIP followed by rapid cooling
- Cumulative thermal cycling that accelerates compression set and material degradation
Some FDA-compliant materials lose compliance at elevated temperatures due to accelerated degradation or increased migration rates. Material selection must be validated against the highest temperature the seal will experience, not just average operating conditions.
Cleaning and Sterilization Methods
CIP and SIP protocols impose chemical and thermal stress that often exceeds food contact conditions:
- CIP (Clean-In-Place) uses caustic chemicals (sodium hydroxide), acids (nitric acid, phosphoric acid), and oxidizers (hydrogen peroxide, peracetic acid)
- SIP (Steam-In-Place) exposes seals to high-temperature steam (250°F-280°F) for extended periods
Materials must resist not just the food product but also the cleaning agents used between production runs. Material-by-material, the tradeoffs look like this:
- EPDM excels in caustic and steam environments
- FKM handles acids and oxidizers but can degrade in superheated steam
- Nitrile resists oils but fails in ozone-generating environments
Mechanical Duty and Seal Design
Compliance addresses safety, but mechanical properties determine seal reliability:
- Dynamic vs. static sealing: Dynamic applications require abrasion resistance and low friction
- Pressure ratings: High-pressure applications need materials with adequate tensile strength
- Compression set resistance: Repeated thermal cycling accelerates permanent deformation
Some highly compliant materials lack mechanical robustness. Soft silicone compounds offer excellent temperature range and non-stick properties but provide inadequate sealing force in high-pressure applications or poor abrasion resistance in dynamic seals.
Range of FDA 21 CFR and NSF 61 Approved Materials
Not every elastomer qualifies for food contact — only specific compounds meet FDA 21 CFR and NSF 61 requirements. The five main material families covered below each carry distinct trade-offs in temperature range, chemical compatibility, and mechanical performance, so matching the right material to your application conditions matters as much as the compliance certification itself.
EPDM (Ethylene Propylene Diene Monomer)
EPDM is the most widely used food-grade elastomer. It handles aqueous and alcoholic foods well, tolerates steam reliably, and holds up against both acids and caustic cleaners — making it the default choice for most water-based food processing environments.
Key specifications:
- Operating range: -40°F to +275°F (standard compounds), up to +350°F with peroxide curing
- Compliance: FDA 21 CFR 177.2600 and NSF 61 certified grades available from major compounders
- Strengths: Exceptional steam resistance, excellent for hot water, caustic cleaners, and polar solvents
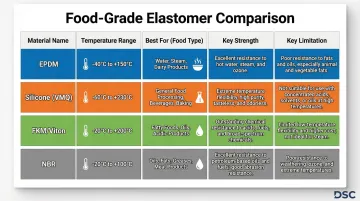
Limitations: Poor resistance to oils, fats, and petroleum-based products. EPDM swells and degrades in fatty food contact, making it unsuitable for applications involving cooking oils, animal fats, or mineral oils.
Silicone (VMQ)
Silicone offers the widest temperature range among food-grade elastomers, excellent flexibility, non-stick surface characteristics, and odorless/tasteless properties that prevent flavor transfer.
Key specifications:
- Operating range: -85°F to +400°F
- Compliance: FDA 21 CFR 177.2600 and USP Class VI (for pharmaceutical-grade applications)
- Strengths: Broadest temperature range, physiological inertness, excellent for dry foods and baked goods
Limitations: Poor abrasion resistance, low tensile strength, limited suitability for dynamic seals or high-pressure applications. Silicone is not recommended for fatty food contact or applications requiring high mechanical strength.
Fluorocarbon (FKM/Viton)
When an application involves fatty foods, aggressive CIP chemicals, or sustained high temperatures, FKM is the go-to material. Its resistance to oils, fats, acids, and strong cleaning agents outperforms every other common food-grade elastomer.
Key specifications:
- Operating range: +15°F to +400°F
- Compliance: FDA 21 CFR 177.2600 and NSF 61 available in specifically compounded grades
- Strengths: Exceptional chemical resistance, excellent for fatty foods, handles aggressive CIP chemicals
Critical warning: Not all FKM grades are food-safe—only specifically compounded versions meet FDA requirements. FKM is susceptible to dehydrofluorination in hot water and steam, potentially releasing hydrogen fluoride. Avoid FKM in steam applications or hot water systems above 200°F.
Nitrile (NBR/Buna-N)
NBR provides an economical option with excellent oil and fat resistance, good abrasion resistance, and tensile strength suitable for dynamic seals in fatty food processing.
Key specifications:
- Operating range: -25°F to +220°F
- Compliance: FDA 21 CFR 177.2600 certified grades available
- Strengths: Excellent oil/fat resistance, good mechanical properties, cost-effective
Limitations: Poor resistance to ozone, UV, and steam. Not recommended for long-term outdoor exposure or high-temperature steam applications. NBR degrades in ozone-generating environments and loses properties in hot water above 200°F.
Specialty and Emerging Materials
Three additional compounds address application gaps that standard elastomers can't cover:
- HNBR (Hydrogenated Nitrile): Operates from -30°F to +300°F with better chemical resistance than standard NBR — suited for high-temperature CIP cycles and hot water wash-down environments
- FVMQ (Fluorosilicone): Pairs silicone's flexibility and temperature range with improved fuel and solvent resistance, useful in alcoholic beverage or flavor compound contact
- Metal-detectable compounds: Incorporate detectable particles while maintaining FDA compliance — essential for HACCP programs using metal detectors or X-ray inspection systems
Key Technical Properties Defining Compliance Performance
Compliance certification addresses safety, but material properties determine operational reliability and service life.
Migration and Extractables Testing
Migration refers to the transfer of chemical substances from the o-ring into the food product. Extractables are substances pulled out under simulated use conditions.
Testing protocols simulate food contact using standardized food simulants:
- Aqueous simulant: Distilled water or 3% acetic acid
- Acidic simulant: 3% acetic acid or citric acid solution
- Alcoholic simulant: 10% ethanol
- Fatty simulant: n-hexane, olive oil, or synthetic triglycerides
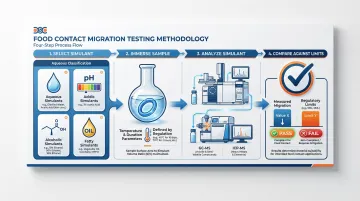
Materials are immersed in these simulants at elevated temperatures for extended periods — typically 7 hours at reflux temperature. The simulant is then analyzed using GC-MS (for volatile organic compounds) or ICP-MS (for trace metals) to quantify leachable substances.
Chemical Compatibility and Resistance
FDA/NSF compliance does not guarantee compatibility with all food products or cleaning agents. Material-specific resistance charts must be consulted to verify compatibility with:
- Cleaning chemicals: Sodium hydroxide (caustic), nitric acid, phosphoric acid, hydrogen peroxide, peracetic acid
- Food ingredients: Citric acid, acetic acid, ethanol, vegetable oils, animal fats, flavor compounds
Always cross-reference your cleaning protocols against material compatibility data — a mismatch here causes premature seal failure, not just reduced service life.
Temperature Stability and Compression Set
Compression set is the permanent deformation remaining after prolonged compression, measured per ASTM D395. High compression set indicates loss of elasticity, which directly affects seal reliability and leak prevention.
Repeated thermal cycling (production → CIP/SIP → production) accelerates compression set. Materials with excellent heat resistance and recovery properties—like peroxide-cured EPDM or fluoroelastomer-grade FKM—maintain sealing force over thousands of cycles.
How FDA 21 CFR and NSF 61 Compliance Is Specified, Measured, and Validated
Compliance is a multi-layered verification process involving material formulation, third-party testing, supplier documentation, and ongoing quality assurance.
Regulatory Documentation and Supplier Certification
Required documentation includes:
- FDA compliance letters confirming conformance to 21 CFR 177.2600
- NSF certification numbers for potable water applications
- Material safety data sheets (SDS) listing full chemical composition
- Ingredient disclosure statements verifying all substances are permitted under applicable regulations
Critical distinction: A supplier stating "FDA compliant" (self-declared) differs significantly from providing third-party test reports and certifications from accredited labs. Third-party certification involves rigorous, ongoing surveillance and facility audits, providing stronger protection against contamination liability than self-declaration alone.
Detroit Sealing Components works with ISO 17025 accredited lab resources and ISO 9001:2015 certified quality system for independent material testing — so customers receive compliance documentation tied to traceable test data, not just supplier declarations.
Migration Testing and Analytical Methods
Migration testing involves immersing o-ring samples in food simulants at specified temperatures and durations, then analyzing the simulant for extractable substances:
- GC-MS (Gas Chromatography-Mass Spectrometry) identifies and quantifies volatile organic compounds migrating from the elastomer
- ICP-MS (Inductively Coupled Plasma-Mass Spectrometry) detects trace metals — lead, zinc, cadmium — at parts-per-billion concentrations
NSF 61 testing includes toxicological evaluation of leachates, comparing results against Total Allowable Concentration (TAC) limits to ensure no harmful substances exceed safe drinking water thresholds.
Traceability and Batch Control
Each o-ring batch should be traceable to raw material certifications and compliance test reports. Lot traceability enables:
- Demonstrates due diligence during regulatory inspections, with documentation ready on demand
- Isolates affected batches quickly if a contamination issue surfaces
- Confirms that manufactured compounds match the exact formulations submitted for compliance testing
Without lot-level traceability, a single out-of-spec batch can compromise an entire product line — and leave you with no paper trail when regulators ask questions.
Implications of Using Non-Compliant Materials in Food Applications
Using non-compliant materials carries serious consequences ranging from regulatory action to public health risks and brand damage.
Non-compliant materials often contain plasticizers, fillers, or curing agents that migrate into food contact surfaces, causing off-tastes, odors, discoloration, or direct chemical contamination. Extractables can alter flavor profiles, shorten shelf life, or introduce harmful substances into your product stream.
When contamination reaches this point, regulatory consequences follow quickly.
Regulatory and legal consequences:
- FDA warning letters and consent decrees
- Mandatory product recalls affecting entire production runs
- Facility shutdowns until compliance is restored
- Civil penalties and fines
- Liability exposure if contaminated products reach consumers
Financial and reputational impact:
- Recall costs including logistics, disposal, and customer notifications
- Lost production time during facility remediation
- Customer trust erosion and brand damage
- Potential exclusion from supply chains requiring certified food-safe components

A single recall event can cost hundreds of thousands of dollars — far exceeding the upfront investment in verified, compliant sealing materials.
Common Misinterpretations of FDA/NSF Compliance in Practice
Several misconceptions create compliance gaps that expose organizations to risk. - "All EPDM is automatically food-safe" — Compliance depends on compound formulation, not the base polymer alone. EPDM compounded with non-permitted curing agents or fillers fails FDA requirements regardless of polymer type.
- "FDA compliance means NSF 61 approval" — These are separate certifications. FDA 21 CFR 177.2600 addresses food contact through extraction limits; NSF 61 addresses drinking water safety through toxicological evaluation. A material can meet one without satisfying the other.
- "Room temperature testing covers high-temperature applications" — Migration rates increase exponentially with temperature. A material passing extraction testing at 100°F may fail at 200°F due to accelerated migration. Temperature-specific validation is required.
- "Material datasheets are sufficient proof of compliance" — Datasheets provide specification data but don't constitute compliance proof. Verification requires supplier certifications, third-party test reports, and lot-specific compliance documentation.
Conclusion
FDA 21 CFR and NSF 61 compliance is a performance-defining requirement, not a passive checkbox. It demands careful material selection, supplier verification, and ongoing documentation. Choosing the right compliant material requires balancing regulatory requirements with real-world operating conditions:
- Temperature extremes during CIP/SIP cycles
- Chemical exposure from aggressive cleaning agents
- Food type — aqueous vs. fatty products
- Mechanical duty and pressure cycling
Each material family has a defined role. EPDM excels in steam and aqueous applications but fails in fatty foods. FKM handles harsh chemicals and fats but degrades in steam. Silicone offers the widest temperature range but lacks mechanical strength. NBR provides economical fat resistance with tighter temperature limits. Match the material to the application — not the other way around.
Detroit Sealing Components supports food and beverage engineers through this process directly. With an ISO 17025 accredited lab and an ISO 9001:2015 certified quality system, access to hundreds of certified compounds, and deep experience in food industry sealing requirements, DSC helps teams move from material specification to validated, documented compliance. If you're sourcing compliant O-rings or evaluating compounds for a new application, that's the conversation to start early — before a failed audit or a contamination event makes it urgent.
Frequently Asked Questions
What is the best material for food-grade O-rings?
No single material fits every application. EPDM suits aqueous foods and steam; FKM handles fatty foods and harsh chemicals; silicone covers the widest temperature range for dry and baked goods; NBR offers economical fat resistance at moderate temperatures. The right choice depends on food type, temperature, cleaning method (CIP/SIP), and mechanical demands.
What is a food contact material?
A food contact material is any substance that comes into contact with food during processing, packaging, or storage. FDA 21 CFR regulates these materials to prevent harmful substance transfer and protect food safety, taste, and odor. Compliance requires approved ingredients and extraction testing to confirm migration limits are not exceeded.
Is silicone safe for food contact and what does FDA 21 CFR 177 say?
Properly compounded silicone meets FDA 21 CFR 177.2600 for repeated food contact, with a working range of -85°F to +400°F. It must be formulated with approved ingredients. Silicone is well-suited for dry foods and baked goods but should be avoided where high abrasion or fatty food contact requires stronger oil resistance.
Is Viton O-ring material food safe and NSF approved?
Viton (FKM) can meet FDA 21 CFR 177.2600 and NSF 61 standards, but only when specifically compounded and certified for food contact—not all FKM grades qualify. It performs well in fatty food and harsh chemical environments. Avoid it in steam or hot water above 200°F, where dehydrofluorination can release hydrogen fluoride.
Are nitrile rubber O-rings food grade?
NBR can be formulated to meet FDA 21 CFR 177.2600, making it a cost-effective option for fatty food applications at moderate temperatures (maximum 220°F). It is not suitable for steam sterilization or outdoor exposure. Approved compounding is required — not all nitrile grades meet food-contact standards.
Is EPDM rubber food grade?
EPDM meets FDA 21 CFR 177.2600 when properly compounded and is a strong choice for dairy, beverages, and steam sterilization — peroxide-cured grades handle steam up to 350°F and resist the acids and caustics used in CIP protocols. It is not suitable for fatty foods or petroleum-based products, where swelling and degradation occur.


